what factors cause water molecules to sink and flow along the bottom of the ocean
Turbidity, Full Suspended Solids & Water Clarity
What are Total Suspended Solids?
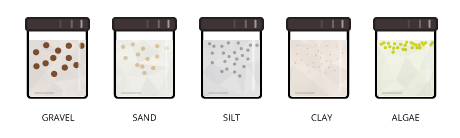
Total suspended solids (TSS) are particles that are larger than ii microns institute in the water cavalcade. Annihilation smaller than 2 microns (boilerplate filter size) is considered a dissolved solid. Most suspended solids are made upwardly of inorganic materials, though bacteria and algae can also contribute to the total solids concentration 3.
These solids include anything drifting or floating in the h2o, from sediment, silt,and sand to plankton and algae 1. Organic particles from decomposing materials can also contribute to the TSS concentration. As algae, plants and animals decay, the decomposition process allows small organic particles to interruption away and enter the h2o column as suspended solids 17. Even chemic precipitates are considered a course of suspended solids twenty. Total suspended solids are a meaning gene in observing water clarity iii. The more than solids present in the water, the less articulate the h2o will be.
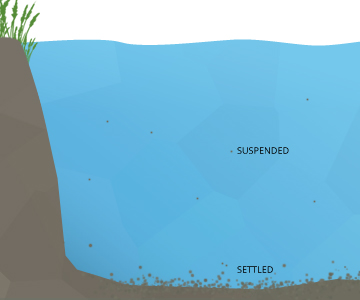
Some suspended solids tin can settle out into sediment at the bottom of a trunk of h2o over a period of fourth dimension 3. Heavier particles, such as gravel and sand, often settle out when they enter an area of low or no water menses. Although this settling improves water clarity, the increased silt tin smother benthic organisms and eggs 1. The remaining particles that do not settle out are chosen colloidal or nonsettleable solids 4. These suspended solids are either too small or as well lite to settle to the bottom.
Settleable solids are also known as bedded sediments, or bedload 37. These sediments can vary from larger sand and gravel to fine silt and clay, depending on the menstruation rate of h2o. Sometimes these sediments can move downstream even without rejoining the suspended solids concentration. When settleable solids are moved along the bottom of a body of water by a strong menstruum, it is called bedload transport 7.
What is Turbidity?

Turbidity is an optical determination of water clarity 1. Turbid water will appear cloudy, murky, or otherwise colored, affecting the physical look of the h2o. Suspended solids and dissolved colored material reduce water clarity by creating an opaque, hazy or muddy appearance. Turbidity measurements are often used as an indicator of h2o quality based on clarity and estimated total suspended solids in water.
The turbidity of water is based on the amount of light scattered by particles in the water cavalcade two. The more particles that are present, the more lite that will be scattered. Every bit such, turbidity and total suspended solids are related. However, turbidity is not a direct measurement of the total suspended materials in h2o. Instead, as a mensurate of relative clarity, turbidity is oft used to signal changes in the full suspended solids concentration in water without providing an verbal measurement of solids 1.

Turbidity can come from suspended sediment such as silt or clay, inorganic materials, or organic matter such as algae, plankton and decaying material. In improver to these suspended solids, turbidity tin also include colored dissolved organic matter (CDOM), fluorescent dissolved organic matter (FDOM) and other dyes 14. CDOM is likewise known as humic stain. Humic stain refers to the tea colour produced from decomposable plants and leaves underwater due to the release of tannins and other molecules.
This discoloration is oftentimes found in bogs, wetlands or other water bodies with high amounts of decaying vegetation in the water. CDOM can cause water to appear red or brown, depending on the blazon of plants or leaves present. These dissolved substances may exist also small to be counted in a suspended solids concentration, but they are nevertheless part of a turbidity measurement as they affect water clarity.
What is Water Clarity?

H2o clarity is a concrete feature divers by how clear or transparent water is. Clarity is determined by the depth that sunlight penetrates in h2o ix. The further sunlight can attain, the higher the water clarity. The depth sunlight reaches is also known equally the photic zone. The clearer the h2o, the deeper the photic zone and the greater the potential for photosynthetic product 10. The photic zone (and thus h2o clarity) has a maximum depth of 200 thou based on the low-cal absorption properties of h2o.
Water clarity is directly related to turbidity, as turbidity is a measure of water clarity. The transparency of water is affected by the corporeality of sunlight available, suspended particles in the water column and dissolved solids such as colored dissolved organic material (CDOM) present in the water.
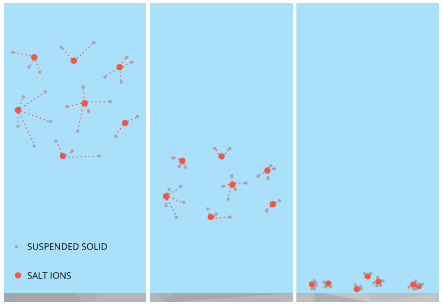
Salinity likewise affects water clarity 10. This is due to the result of common salt on the aggregation and settling velocity of suspended particles. In other words, common salt ions collect suspended particles and demark them together, increasing their weights and thus their likelihood of settling to the bottom. Due to this mechanism, oceans and estuaries tend to accept a higher clarity (and lower average turbidity) than lakes and rivers 10. These marine environments likewise take a higher rate of sedimentation as solids are pulled out of the water column to the seafloor.
Turbidity vs Suspended Solids – What is the divergence?
Turbidity and total suspended solids refer to particles nowadays in the h2o cavalcade. Turbidity and water clarity are both visual properties of water based on lite scattering and attenuation. All 3 parameters are related to particles in the water column, whether straight or indirectly.
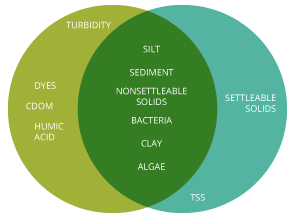
Turbidity is adamant by the corporeality of light scattered off of these particles 6. While this measurement can so be used to estimate the total dissolved solids concentration, it will non be exact. Turbidity does not include any settled solids or bedload (sediment that "rolls" along the riverbed) 7. In addition, turbidity measurements may be afflicted by colored dissolved organic matter v. While this dissolved matter is not included in TSS measurements, it can crusade artificially low turbidity readings as it absorbs calorie-free instead of scattering it 8.
Total suspended solids, on the other hand, are a full quantity measurement of solid material per book of water 6. This means that TSS is a specific measurement of all suspended solids, organic and inorganic, past mass. TSS includes settleable solids, and is the straight measurement of the total solids present in a water body. As such, TSS can exist used to calculate sedimentation rates, while turbidity cannot 1,vi.
Water clarity is strictly relative to sunlight penetration. While this is ordinarily determined past the amount of suspended solids in h2o, it can too be affected by CDOM and other dissolved solids 10. H2o clarity is the nigh subjective measurement of the these three parameters, equally it is ordinarily determined by human ascertainment 5.

Why are Turbidity and Full Suspended Solids Important?

Turbidity and TSS are the most visible indicators of water quality. These suspended particles can come up from soil erosion, runoff, discharges, stirred bottom sediments or algal blooms ane. While it is possible for some streams to have naturally loftier levels of suspended solids, clear water is unremarkably considered an indicator of healthy water 9,14. A sudden increase in turbidity in a previously clear body of water is a cause for business concern. Excessive suspended sediment can impair water quality for aquatic and human life, impede navigation and increase flooding risks seven.
Water Chemistry
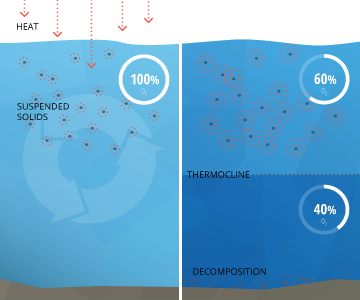
In terms of h2o quality, loftier levels of total suspended solids will increase water temperatures and subtract dissolved oxygen (Exercise) levels 1. This is considering suspended particles absorb more heat from solar radiation than water molecules volition. This heat is and then transferred to the surrounding water by conduction. Warmer water cannot hold as much dissolved oxygen as colder h2o, so Practice levels will drop 13. In addition, the increased surface temperature tin cause stratification, or layering, of a body of water 3. When h2o stratifies, the upper and lower layers do not mix. Every bit decomposition and respiration oft occur in the the lower layers, they can get too hypoxic (low dissolved oxygen levels) for organisms to survive.
Photosynthesis Production
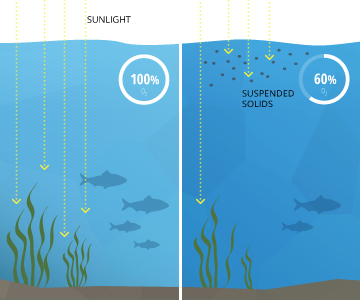
Turbidity tin also inhibit photosynthesis by blocking sunlight. Halted or reduced photosynthesis ways a decrease in institute survival and decreased dissolved oxygen output nine. The higher the turbidity levels, the less light that can reach the lower levels of h2o. This reduces plant productivity at the bottom of an ocean, lake or river fifteen. Without the needed sunlight, seaweed and bay grasses below the water'south surface will non exist able to continue photosynthesis and may die xvi.
Underwater vegetation dice-off has two main effects. Start, as photosynthetic processes subtract, less dissolved oxygen is produced, thus further reducing Exercise levels in a body of water 13. The subsequent decomposition of the organic cloth can driblet dissolved oxygen levels even lower. Second, seaweed and underwater plants are necessary food sources for many aquatic organisms. As they die off, the amount of vegetation available for other aquatic life to feed on is reduced. This can crusade population declines up the food chain 21.
Erosion

An increase in turbidity can also indicate increased erosion of stream banks, which may have a long-term issue on a body of h2o 3. Erosion reduces habitat quality for fish and other organisms. In terms of water clarity, reduced light penetration due to suspended sediment can obscure aquatic organisms' vision, reducing their power to find food 17. These suspended particles can also clog fish gills and affect growth rates thirteen.
Erosion can contribute to shallower, filled-in lakes and streams as some of the suspended particles settle out 15. These settleable solids can suffocate benthic organisms and fish eggs ane. In add-on, the sediment may smother insect larvae and other fish nutrient sources 13. When this occurs in rivers and channels, the increased sediment loads tin can reduce navigability for ships and boats 7. In cases of excessive sedimentation, settleable solids from erosion and runoff tin even halt freight passage completely.
Contagion

Pollutants such as dissolved metals and pathogens can attach to suspended particles and enter the water ii. This is why an increase in turbidity can often indicate potential pollution, not just a decrease in water quality. Contaminants include bacteria, protozoa, nutrients (e.g. nitrates and phosphorus), pesticides, mercury, lead and other metals 17. Several of these pollutants, peculiarly heavy metals, tin be detrimental and often toxic to aquatic life 26. The addition of nutrients can encourage the development of harmful algal blooms.
When the suspended solids concentration is due to organic materials, particularly sewage effluent and decaying organic matter, the presence of bacteria, protozoa and viruses are more probable. These organic suspended solids are as well more than likely to subtract dissolved oxygen levels equally they are decomposed 19.
Human Concerns
These microbes and heavy metals can impact not just aquatic organisms, but drinking h2o too 2. Organic suspended solids, such as decomposing matter or sewage effluent often naturally include high levels of microorganisms such as protozoa, leaner and viruses 19. Such pathogens contribute to waterborne diseases like cryptosporidiosis, cholera and giardiasis 25. Turbid water, whether due to organic or inorganic textile, cannot be easily disinfected, every bit the suspended particles volition "hide" these microorganisms twenty.
In a lake or river, turbidity may also reduce visibility of underwater structures such as logs or large boulders, negatively affecting a water body'due south recreational use nineteen. In industrial processes, turbidity can contribute to clogged tanks and pipes xx. The particles can also scour machines, potentially damaging them.
What Contributes to Suspended Solids?

Suspended solids in a torso of water are frequently due to natural causes. These natural solids include organic materials such as algae, and inorganic materials such as silt and sediment. Some algae, such every bit phytoplankton, are regular occurrences, especially in the ocean. Inorganic materials can hands get suspended due to runoff, erosion and resuspension from seasonal water period. However, when suspended solids exceed expected concentrations, they can negatively bear upon a body of h2o. Excess over background amounts are often attributed to human influence, whether direct or indirectly 37. Pollution may contribute to either organic or inorganic suspended solids, depending on the source. Algae, sediment and pollution will affect water quality in different means depending on the quantity nowadays.
Algae

Algae are plantlike, photosynthesizing organisms that can thrive in both freshwater and saltwater 11. These organisms come in unlike sizes, from microscopic phytoplankton to behemothic sea kelp forests 12. Both the phytoplankton and seaweed forms of algae will swallow nutrients in the h2o and can increment dissolved oxygen levels through photosynthesis. When they die, however, the organic material is decomposed by microbes in the water cavalcade. This decomposition process can decrease dissolved oxygen levels to beneath normal levels 42.
Seaweed and kelp are found rooted to the seafloor, only phytoplankton and other micro-algae tin can be found at the water's surface or throughout the water column xi. In particular, cyanobacteria, or blue-dark-green algae, accept floating mechanisms that keep them at the surface, blocking sunlight from the water nineteen. These phytoplankton contribute to the full suspended solids concentration, while rooted vegetation or attached streambed-mat forms of algae do not. However, if these rooted algae get detached (usually when the algae dies or if it is forcefully removed), then their mass becomes function of the suspended solids measurement 6.

The most obvious examples of algae's contribution to turbidity are institute in algal blooms eleven. An algal bloom occurs when an excessive amount of algae grows quickly across the surface of a body of h2o. These blooms ordinarily occur due to an influx of nutrients such as nitrogen and phosphorus due to agricultural runoff or decomposition, though warmer water temperatures and longer daylight hours also contribute to their growth 15. Floating algal blooms tin can cake sunlight, release toxins, and deplete oxygen levels in a body of water xi.
While some algae growth occurs naturally (frequently seasonally), excessive growth is oftentimes attributed nutrient pollution.Turbidity monitoring can be used to determine if an increase in suspended solids is natural or due to agricultural runoff 6.
Sediment: Runoff and Erosion
Sediment is comprised of any solid material that tin exist transported by water, wind or ice 23. Information technology is usually defined equally the soil particles (including silt, clay and sand) that are deposited on the bottom of a body of water 21. These particles are usually classified by size from smallest (dirt is less than 0.00195 mm in diameter) to largest (coarse sand can be upwardly to ane.v mm) 19. Silt falls in between, ranging from 0.0049 to 0.047 mm.

In areas of high flow, even rocks tin exist considered sediment as they are deposited in h2o 22. Nevertheless, non all sediment is suspended. The amount and size of suspended sediment is dependent on water flow 17. The faster the menstruation, the larger the particle that can exist suspended. College flow rates can too support a higher concentration of suspended solids. Particles larger than 0.five mm usually settle out as water flow decreases 19. Most of the suspended sediment that remains (colloidal solids) consists of fine sand, silt, and dirt nineteen.
The majority of suspended sediment present in water bodies comes from runoff and erosion six. If the land surrounding a torso of water has only thin vegetation, the topsoil tin easily be washed away into the water three. Highly vegetated areas will absorb well-nigh of the runoff, keeping the body of water clearer.

In improver to collecting suspended particles from runoff, rivers and streams tin slowly erode soft riverbanks due to the constant water period. An increase in river volume and menstruation (due to rain or other causes) can increase the charge per unit of erosion eighteen. On the other side of the spectrum, bedrock-based streams may not have much sediment available to append. The local geology will decide natural turbidity levels based on normal menses rates, soil type, land structure and vegetation 6. If the surrounding land is contradistinct by agriculture, construction or other soil-disturbing use, information technology tin accelerate erosion and runoff, increasing turbidity 3.
Pollution

Any potentially harmful substance that is added to the surroundings by humans, whether directly or indirectly, is considered pollution 29. This can vary from leaner riding along on a sewage plant discharge, to coal and iron ore particulates that bladder in from a mining zone. If these pollutants are larger than 2 microns, they volition contribute to the total suspended solids concentration.
Some of the more than common suspended solid pollutants are pathogens (bacteria, protozoa, helminths), microbeads (from exfoliating soaps), wastewater effluent, sewage, airborne particulates, and route particles (east.grand. asphalt and tire flecks). Colored wastewater discharge and dyes are pollutants that volition impact turbidity, only non suspended solids.
Nutrients like nitrate and phosphorus are often considered pollutants, but every bit they are a dissolved substance, they do not contribute directly to the suspended solids concentration thirty. Instead, they are an indirect contributor as they fuel algal blooms, which do bear upon TSS and turbidity.
These dissolved nutrients, along with dissolved metals, chemicals, and refractory organics, will bear on the quality of a body of h2o 29. Nitrate and phosphorus can crusade eutrophication (excessive plant and algae growth) which in turn causes low dissolved oxygen levels due to plant respiration and microbial decomposition. Refractory organics are oftentimes carcinogenic, while heavy metals and other chemicals can be toxic to aquatic organisms 29.
While these contaminants can enter the water as a dissolved substance, many of them ride along on grains of soil or other larger pieces of pollution (e.m. paint flecks or asphalt particles)29. When this is the case, they can be picked up in suspended sediment samples. Chemical dyes will bear upon turbidity readings as the colored molecules will touch on light absorption, simply they will not exist included in a suspended solids measurement.
Factors that Influence Turbidity
Suspended solids can be comprised of organic and inorganic materials such as sediment, algae, and other contaminants. However, in that location are specific factors that can affect turbidity levels in a body of water. These are water flow, signal source pollution, land use and resuspension.
Water Catamenia and Weather
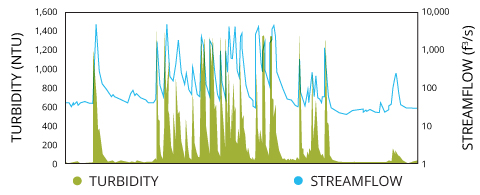
Turbidity and water menstruum are causally related xviii. High flow rates go on particles suspended instead of letting them settle to the bottom. Thus in rivers and other naturally-occurring high flow environments, turbidity can be a abiding presence 2. In these areas, it is important to monitor for changes in turbidity at the same point each time to ensure that the data is not affected by a lower or college water velocity 18.
Weather, particularly heavy rainfall, also affects water flow, which in plow affects turbidity. Rainfall tin can increase stream volume and thus stream flow, which can resuspend settled sediments and erode riverbanks one.
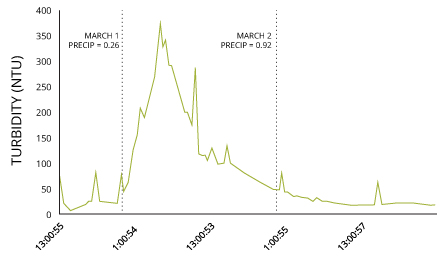
Rain can besides directly increase the level of total suspended solids through runoff. Every bit water flows over a surface, it can pick up particles and deposit them in a torso of water two. Runoff can also wash away topsoil, and contribute to riverbank erosion 3. If the flow rate increases enough, information technology can resuspend bottom sediments, further raising TSS concentrations 2.
In areas of dry, loose soil or earth-disturbed sites (e.g. mining or structure areas), wind tin blow dust, sediment and other particles into the water 29. The addition of new particles will increment the suspended solids concentration. However, air current will generally not increment turbidity levels in the water alone. In wave-dominated estuaries and littoral areas, turbidity is naturally low 41. In comparing, tidal areas, where the h2o flow is stiff enough to resuspended lesser sediments, have high natural turbidity levels. Wind-driven turbidity increases but occur in shallow zones where waves are tall enough to resuspend sediment 40. Tides, wind, and rain can influence turbidity levels due to their effect on water menstruation and introduced sediment loads 9.
Tributaries can also alter turbidity. When a freshwater stream or river enters a saltwater estuary, the change in water menstruum can cause turbidity levels to increase. This mixing area is often called a turbidity maximum zone 16. These zones tend to take picayune aquatic vegetation due to the high suspended solids concentrations. Estuaries are often subject to tidal influences as well, which tin pull in sand and sediment from the shoreline and resuspended bottom sediments 16.

Betoken-Source Pollution
If pollution can be tracked to a single, identifiable source, it is considered point-source pollution 34. Betoken-source pollution tin can increase turbidity through the addition of suspended solids and colored effluent (wastewater) to a ocean. For water quality, mutual examples include discharge pipes from factories and wastewater handling plants. In improver, farms can also fall under the category of point-source pollution 33. These sources can release harmful pathogens (bacteria) and chemicals into the water, in addition to suspended solids.

Many factories, wastewater treatment plants, and sewage treatment plants belch effluent into local water bodies or sewer systems. Sometimes this water is treated or filtered before it is discharged, but sometimes it is not 33. The EPA has created several guidelines for effluent discharge, but they are all based on the technology used, and not the final impact on the local h2o trunk 32. While virtually wastewater handling plants include a settling flow in the treatment process, this does non bear on colloidal (nonsettleable) solids 13. When this wastewater is discharged, these suspended solids may still be present unless treated with additional filters. In add-on, colored effluent cannot exist trapped by a filter. While dyes and colored dissolved organic material (CDOM) are non included in a suspended solids measurement, they will contribute to turbidity readings due to their effects on calorie-free absorption.
Farms that are identified as point sources often allow fertilizer and animal waste matter to enter local bodies of water. Most agronomical pollution is due to runoff, and non a specific discharge. While this runoff is not intentional, it can be detrimental to water quality as these pollutants are untreated 33. Fauna wastes can increase pathogen concentrations in the water, while the fertilizer can contribute to eutrophication and excessive algal growth.
Land Use

A major factor in increased turbidity and total suspended solids concentrations is due to land use. Construction, logging, mining and other disturbed sites have an increased level of exposed soil and decreased vegetation 17. Agricultural areas are also considered disturbed areas later they are tilled 18. Land evolution, whether it is agricultural or structure, disturbs and loosens soil, increasing the opportunities for runoff and erosion xv. The loosened soils caused past these sites tin can so exist carried away by current of air and rain to a nearby body of water.
This leads to an increase in runoff rates, causing erosion and increased turbidity in local streams and lakes six. Settleable solids in the runoff can be deposited on the bottom of a lake, river or ocean, damaging benthic habitats 15. Erosion due to land utilise is considered a non-betoken source of turbidity. The use of silt fences and sedimentation basins at structure sites can prevent soils from reaching nearby water sources 13.
In addition to increasing turbidity levels through suspended sediment, agricultural runoff often includes nutrients as well. Due to the presence of these nutrients, this runoff can fuel the growth of algal blooms ix. These effects tin be seen in local streams, lakes, and even estuaries like the Chesapeake Bay. Water quality can be affected anywhere that these nutrients and sediments are carried. No-till farming practices can reduce the potential for erosion and aid maintain nearby h2o quality 13.

Sediment- and pollutant-filled runoff can also occur in urban areas. When it rains, soil, tire particles, debris and other solids can get washed into a water system 17. This often occurs at a high catamenia charge per unit due to the corporeality of impervious surface areas (eastward.g. roads and parking lots). H2o cannot penetrate these surfaces, so sediment cannot settle out 17. Instead, the stormwater runoff flows right over the pavement, carrying the suspended solids with it. Even in areas with storm drains, these drains ordinarily lead directly to a local h2o source without filtration 21. To minimize the pollution and turbidity caused by urban runoff, stormwater retention ponds tin be constructed 13. These basins let suspended particles to settle before water drains downstream 28.
Resuspension
Even carp and other bottom-feeding fish can contribute to increased turbidity levels 17. As they remove vegetation, sediment can become resuspended in the water. Sediment at the bottom of a ocean can be stirred up by shifting water catamenia, bottom-feeding fish, and anthropogenic causes such as dredging. Dredging projects, which remove congenital-up sediment in navigation channels, are a major source of resuspended sediments in the surrounding water iii. Dredging can cause high turbidity levels as information technology disturbs large amounts of settled sediment in a relatively short period of time. These stirred-up particles are mostly silt and sand. When they resettle, they tin alter habitats, smother fish eggs and suffocate bottom-dwelling organisms.

TSS and Turbidity Units
Full suspended solids, as a measurement of mass are reported in milligrams of solids per liter of h2o (mg/L) eighteen. Suspended sediment is also measured in mg/50 36. The most authentic method of determining TSS is by filtering and weighing a h2o sample 44. This is often time consuming and hard to measure accurately due to the precision required and the potential for error due to the cobweb filter 44.
Turbidity, on the other hand, is most often measured with a turbidity meter. Turbidity is reported in units called a Nephelometric Turbidity Unit (NTU), or a Jackson Turbidity Unit of measurement (JTU) 6. The JTU was the original turbidity unit based on the visibility of candlelight in a tube (Jackson Candle Turbidimeter) 43. Even so, this method is considered out of engagement and inaccurate in comparison to newer methods.
While some organizations consider the two units to be approximately equal, at that place are some specific differences 20. In item, NTU is more precise and has a wider range (JTU cannot measure above 25 JTU/NTU) 43. In improver NTU is the standard unit of many broadband output (400-680 nm wavelength) turbidit meters. Nephelometric refers to the measurement technology used. This technology method requires the photodetector in the meter to be placed at a 90 degree angle from the illumination source fourteen. As light bounces off the suspended particles, the photodetector tin can measure the scattered light.
The USGS likewise suggests the utilize of the Formazin Nephelometric Unit (FNU) if a turbidity meter only has a monochrome/infrared output, as opposed to the white/broadband output fourteen. This applies to instruments that are in compliance with the European drinking-water protocol, including near submersible turbidity meters 34. Both NTU and FNU will prove equal measurements when calibrating as they both employ nephelometric engineering, simply may operate differently in the field due to the dissimilar low-cal source 14. Turbidity meters that use FNU units are able to compensate for dissolved colored materials (such as humic stain), while NTU turbidity meters cannot 14.

Water clarity, when not measured in terms of turbidity, is measured by Secchi depth i. This measurement is based on the depth that a black and white Secchi disc can be lowered into a trunk of h2o. At the bespeak visibility is lost, the depth of the disc is recorded, and is known equally the Secchi depth 16. High Secchi depths represent with low turbidity levels, while low Secchi depths are associated with high levels of suspended solids. This method is generally only useful in oceans, lakes and deep, low-flow rivers. In marine environments, a larger solid white disc is often used, while some shallower lakes use a black disc and take a horizontal measurement 39.
Due to the effects of salt on suspended sediment, ocean clarity is oft much higher than lake or river clarity. Near Secchi disc records reach around 65-eighty thousand 39. Water clarity has a theoretical limit of 200 one thousand, based on light penetration and calculations with distilled and ultrapure water ten. Still, nigh Secchi discs are not large enough to be seen at that depth.
In shallower streams, a Secchi tube tin can exist used 38. A Secchi tube is usually one meter long and is filled with nerveless water. A small Secchi disc is then lowered into the tube and read at the point of disappearance, just as information technology is in a larger body of h2o 38.
Turbidity Meters and Measurements
Regardless of whether readings are in NTU, FNU or other less common units, information technology is important to notation that a turbidimeter'due south optical design volition touch turbidity readings 14. As turbidity is a measurement of calorie-free scatter, the placement and designs of the detectors with the meter tin can influence the readings. This simply means that raw data from two dissimilar turbidity meters cannot be directly compared without an established relationship betwixt them xiv. Turbidity readings tin can vary based on wavelengths emitted, lite source instability, high particle density or due to the presence of colored dissolved or suspended fabric. The more detectors present in a turbidimeter, the less variability there will be in measurements fourteen.
Typical Levels

In most situations, a total suspended solids concentration below 20 mg/L appears clear, while levels over 40 mg/50 may begin to announced cloudy 13. In comparison, a turbidity reading below 5 NTU appears clear, while a reading of 55 NTU will commencement to await cloudy and a reading over 500 NTU volition appear completely opaque two.
It is of import to note that this is dependent on the size and nature of the suspended solids. Typical turbidity and TSS levels are difficult to quantify due to their natural variation by flavor, local geology, h2o menses and weather events. During a low-flow menstruum, well-nigh rivers and lakes are fairly articulate with a turbidity reading beneath 10 NTU. These readings can easily jump into the hundreds due to runoff during a rainstorm, snowmelt or a dredging project 1.
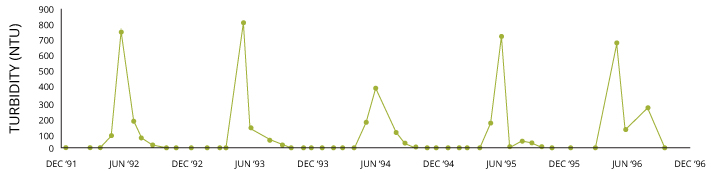
In general, marine environments have lower turbidity levels than freshwater sources 10. The salinity of the sea or estuary volition cause the the suspended solids to aggregate, or combine. As the aggregate weight increases, the solids begin to sink and will settle on the seafloor ten. This effect offers greater water clarity than is available in about lakes and rivers. The higher the salinity, the greater the effect x. Withal, in tidal zones, a turbidity maximum may occur due to the constant resuspension of these settled solids 16. Freshwater sources may as well carry out boosted suspended particles into the delta.

Every bit the concentrations of total suspended solids are hard to measure and predict, nearly states exercise not have a set standard. Even the National University of Sciences only recommends that "TSS should not reduce low-cal penetration by more than 10%" 3. Kentucky does non take a quantitative standard for acceptable levels of total suspended solids. Instead, they simply country that at that place should be no adverse affects to the body of water or its inhabitants three. Michigan is another case of a state with only a "narrative standard" for total suspended solids and turbidity thirteen. In that location is no ready level or concentration, only a recommendation against unnatural physical properties (eastward.k. turbidity, color, films, floating or suspended solids) in "injurious" quantities thirteen.

Instead, many countries and organizations take established recommended turbidity levels from a baseline of prior measurements. In the instance of drinking water, recommended levels are based on several filtration and disinfection studies 31The Ireland EPA advises treatment plants to take turbidity levels below 0.two NTU, with a mandatory maximum of one NTU for drinking water 31. Co-ordinate to the World Wellness Organization, water for human consumption should have turbidity levels below 1 NTU, though for some regions, up to 5 NTU is allowed if it can be proven to be disinfected xix,20. The American H2o Works Association suggests that a level of five NTU or lower is acceptable for recreation purposes 19. As a land case, the North Carolina code allows upwardly to 10 NTU for trout waters, 25 NTU for non-trout streams and as high as fifty NTU for non-trout lakes.
Other states have determined commanded fluctuations from an established baseline. The land of Washington does not have a standard for TSS, simply it does for turbidity, depending on the body of water. In some streams, turbidity cannot increase past more than 5 NTU from the baseline. For others, turbidity may be allowed to fluctuate by up to 20% 6.
Consequences of Unusual Levels
In addition to to being a warning sign for pollution, suspended solids can harbor pathogens such as bacteria and protozoa 2. These microorganisms attach to the suspended particles, aiding in their transportation and hiding them from disinfectants 2. These pathogens tin infect aquatic or human life if the sediment is non removed.
Algal Blooms
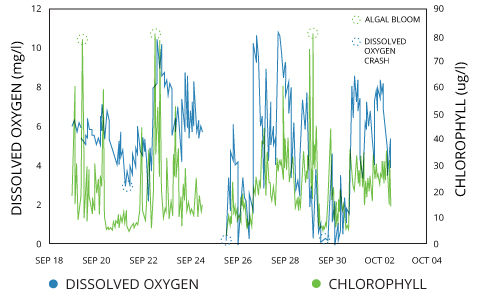
Algal blooms, while initially increasing dissolved oxygen levels, may create hypoxic conditions as they decompose 42. When an algal bloom appears, it blocks sunlight from reaching any submerged vegetation, killing those plants and decreasing the amount of dissolved oxygen produced. And so, when the bloom dies off, microbes swallow more oxygen every bit they decompose the organic cloth. This causes dissolved oxygen levels to plummet even lower, creating hypoxic (low Practice) or even anoxic (no Exercise) weather condition.
Furthermore, some blooms produce toxins that are dissentious to aquatic and human life xvi. These harmful algal blooms include cyanobacteria, cherry-red tide (Karenia brevis) and ciguartera (gambierdiscus toxicus) 27.
Settleable Solids
Settleable solids tin can impair lakes and other water bodies. If sedimentation rates are high, they tin alter and oftentimes destroy fish habitats and spawning beds 37. If eggs or benthic organisms are present, they tin become cached by the sediment and dice. Sediment degradation tin can reduce egg and embryo survival by reducing oxygen supply and crusting over the egg, preventing the embryo from escaping 37.
Every bit sediment build-upwardly increases, the shallower torso of water ways an increased risk of flooding and a decrease in navigability for boats and ships 24. Dredging projects attempt to remove excessive sediment deposits from navigation channels, only this tin can exist just as dissentious to the local fish habitats and spawning beds.
Turbidity
High turbidity levels can diminish visibility and often feeding behaviors, in improver to physically harming aquatic life. The suspended solids may disrupt the natural movements and migrations of aquatic populations thirteen. Fish that rely on sight and speed to catch their prey are especially afflicted by high turbidity levels. These fish often flee areas of high turbidity for new territories 37. For the fish that remain in the turbid surround, suspended sediment tin can begin to physically bear on the fish. Fine sediment tin can clog fish gills and lower an organism's resistance to disease and parasites 37. Some fish may consume suspended solids, causing illness and exposing the fish to potential toxins or pathogens on the sediment. If the consumed sediment does non kill the fish, information technology can alter the organism's blood chemistry and impair its growth 37.
Turbidity volition likewise touch on submerged plant growth. Levels over fifteen NTU are considered detrimental to bay grass growth in estuary zones 16. As turbidity increases, the corporeality of light available to submerged aquatic vegetation (SAV) decreases. Without enough low-cal, photosynthesis will cease, and the SAV will no longer produce dissolved oxygen. In addition to reducing the dissolved oxygen concentration in the h2o, the plants will eventually die forty. As the aquatic vegetation dies off, the organisms that feed on it will also decline due to the reduced food sources available xiii. If turbidity levels remain elevated, the furnishings can be seen up the food chain 37.
Even aquatic life that does non strongly depend on vegetation for survival volition be afflicted by the depression dissolved oxygen levels 40. If these fish and invertebrate cannot escape the anoxic area, they volition die.
Cite This Work
Fondriest Environmental, Inc. "Turbidity, Total Suspended Solids and Water Clarity." Fundamentals of Environmental Measurements. thirteen Jun. 2014. Web. < https://world wide web.fondriest.com/environmental-measurements/parameters/water-quality/turbidity-total-suspended-solids-water-clarity/ >.
Additional Information
- Turbidity Measurement Methods
- Turbidity Meters
- Turbidity Sensors
- Applications
- References
Source: https://www.fondriest.com/environmental-measurements/parameters/water-quality/turbidity-total-suspended-solids-water-clarity/
0 Response to "what factors cause water molecules to sink and flow along the bottom of the ocean"
Post a Comment